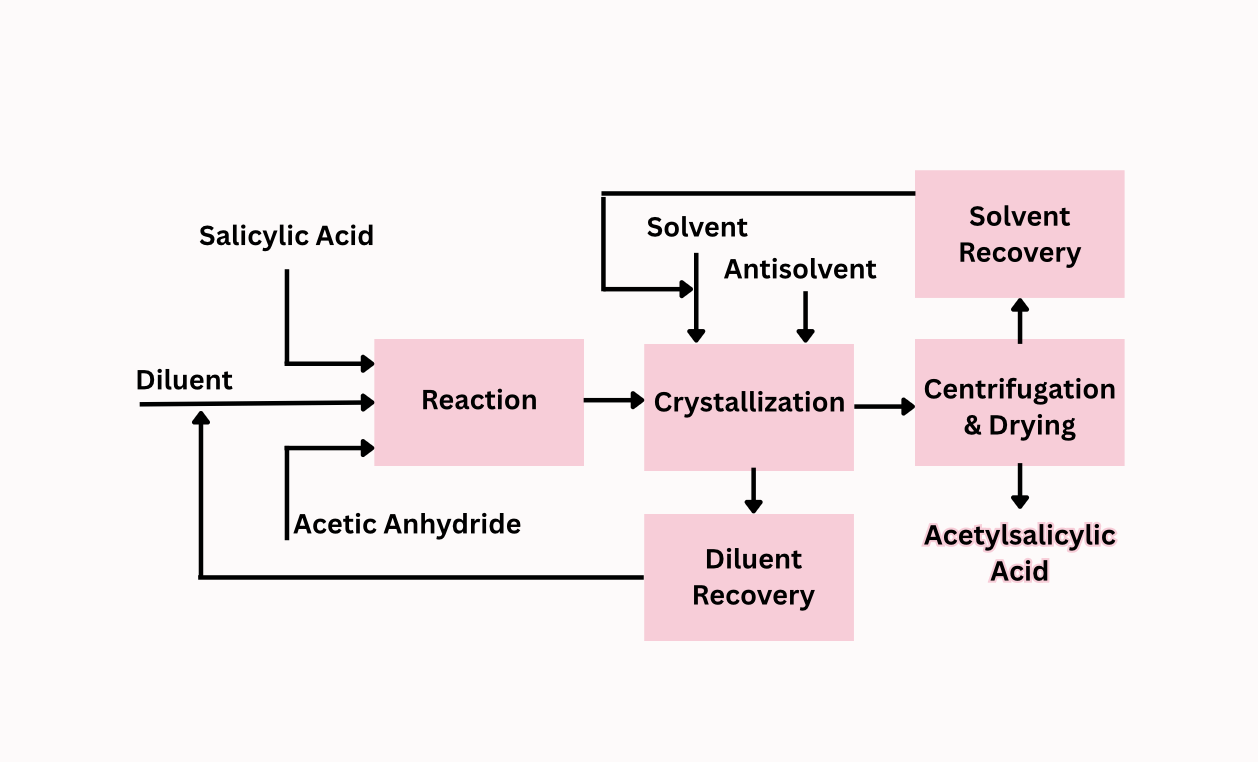
Project Description:
The current process of producing acetylsalicylic acid (aspirin) is outdated and generates hazardous waste. Most pharmaceutical production processes generate anywhere from 25 to 100 kg of waste per kg of product. This waste is a serious environmental and economic concerns, as it contains valuable materials along with toxic materials. The manufacturing process follow FDA and state regulations for drug manufacturing. The feed stream consists of acetic acid combined with salicylic acid and acetic anhydride. The feed reacts in the continuous reactor (PFR) and is subsequently crystallized with a solvent and an anti-solvent. The product is centrifuged and dried to recover the product, acetylsalicylic acid (aspirin). Recovered acetic acid from the crystallization process is recycled into the feed stream. Recovered solvent from the centrifuge and drying process is recycled into the crystallization process. GSK, a pharmaceutical company in Rockville, MD has a manufacturing plant which previous produced aspirin. However due to current sustainability goals and supply chain issues, the product has been halted. GSK plans for waste reductions does not identify by which measures they plan to reduce waste production in their supply chain. Our process is a method GSK can use to reduce waste production in their supply chain.